Reps for Research donations to support lung cancer research

This spring, Hoosier Cancer Research Network Chairman Christopher A. Fausel, PharmD, offered a unique challenge to friends and supporters of HCRN. Combining his interest in strength training and his passion for investigator-initiated research, Fausel invited pledges for every bench-press “rep” he could complete at the Arnold Sports Festival’s 5K Pump and Run event in Columbus, […]
Fisher Award recipient explores immunotherapy targets in circulating tumor cells

Gregory A. Durm, MD, a hematology/oncology fellow at the Indiana University School of Medicine, is helping to break new ground in the fight against cancer by studying circulating tumor cells (CTCs) in lung cancer. CTCs are cells that have broken away from primary tumors and have entered into the bloodstream. CTCs that survive the bloodstream’s […]
HCRN opens PD-1 inhibitor trial for non-small cell lung cancer

Hoosier Cancer Research Network is pleased to announce the opening of a cancer clinical trial for patients with stage III non-small cell lung cancer at select sites throughout the United States. The study, known as LUN14-179, seeks to determine if the use of an investigational drug, Pembrolizumab, following standard chemoradiation treatment, improves the time to […]
HCRN honors investigators with awards

Hoosier Cancer Research Network recently honored two investigators with awards for their contributions to clinical cancer research. Gabriela Chiorean, MD, received the 2014 Danny Danielson Translational Innovation Award. The award was created in 2013 by Donald C. “Danny” Danielson and is granted twice each year through the Walther Cancer Foundation to investigators working in partnership […]
Einhorn reflects on milestone anniversaries

As Hoosier Cancer Research Network celebrates our 30th anniversary, we acknowledge another significant milestone: the 40th anniversary of the discovery of the cure for testicular cancer by HCRN co-founder and board member, Lawrence H. Einhorn, MD. Dr. Einhorn joined the Indiana University School of Medicine in 1973, just one year before his groundbreaking discovery testing […]
Galsky receives Danny Danielson Translational Innovation Award

Hoosier Cancer Research Network, formerly known as Hoosier Oncology Group, recently honored Matthew Galsky, MD, as recipient of the Danny Danielson Translational Innovation Award. The $10,000 award, granted by the Walther Cancer Foundation through the generous support of business and civic leader Donald C. “Danny” Danielson, is given twice each year to investigators working in […]
Matei receives Fisher Young Investigator Award
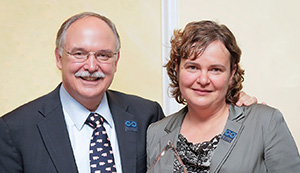
Hoosier Cancer Research Network, formerly known as Hoosier Oncology Group, recently honored Daniela Matei, MD, as the 2014 recipient of the George and Sarah Jane Fisher Young Investigator Award. Dr. Matei received her medical degree in her native Romania before moving to the United States and completed her residency at SUNY at Stony Brook, NY, […]
Matei receives 2011 Research Frontiers Trailblazer Award
Dr. Daniela Matei received the Research Frontiers Trailblazer Award for her discoveries related to the biology of ovarian cancer at the second annual Research Day conference. The Trailblazer award honors outstanding IUPUI researchers who are “showing great promise in becoming nationally and internationally known for their accomplishments in advancing the frontiers of knowledge.”
HCRN honors five during 30th anniversary dinner
Hoosier Cancer Research Network (HCRN) recently marked its 30th anniversary with a dinner and presentation of awards. The event featured comments by HCRN co-founder Patrick J. Loehrer, Sr., MD and chairman of the board Christopher A. Fausel, PharmD, MHA, BCOP. HCRN honored five individuals during the June 19 event in Indianapolis.
HCRN honors five during 30th anniversary dinner
Hoosier Cancer Research Network (HCRN) recently marked its 30th anniversary with a dinner and presentation of awards. The event featured comments by HCRN co-founder Patrick J. Loehrer, Sr., MD and chairman of the board Christopher A. Fausel, PharmD, MHA, BCOP. HCRN honored five individuals during the June 19 event in Indianapolis.
Robin T. Zon, MD, FACP, FASCO, to present Education Session at ASCO

Former Hoosier Cancer Research Network vice-chair Robin T. Zon, MD, FACP, FASCO, will chair an Education Session during the American Society of Clinical Oncology (ASCO) annual meeting in Chicago. Zon, along with Jeffrey S. Abrams, MD, of the National Cancer Institute at the National Institutes of Health, and Nicholas J. Robert, MD, of Virginia Cancer […]
Robin T. Zon, MD, FACP, FASCO, to present Education Session at ASCO

Former Hoosier Cancer Research Network vice-chair Robin T. Zon, MD, FACP, FASCO, will chair an Education Session during the American Society of Clinical Oncology (ASCO) annual meeting in Chicago. Zon, along with Jeffrey S. Abrams, MD, of the National Cancer Institute at the National Institutes of Health, and Nicholas J. Robert, MD, of Virginia Cancer […]
Hoosier Cancer Research Network co-founder elected AACR fellow

Lawrence Einhorn, M.D., the physician-researcher who developed the cure for testicular cancer, has been elected a fellow of the American Association for Cancer Research. Dr. Einhorn, Indiana University Distinguished Professor and a physician-researcher at the Indiana University Melvin and Bren Simon Cancer Center, will be inducted into the American Association for Cancer Research Academy Class […]
Hoosier Cancer Research Network co-founder elected AACR fellow

Lawrence Einhorn, M.D., the physician-researcher who developed the cure for testicular cancer, has been elected a fellow of the American Association for Cancer Research. Dr. Einhorn, Indiana University Distinguished Professor and a physician-researcher at the Indiana University Melvin and Bren Simon Cancer Center, will be inducted into the American Association for Cancer Research Academy Class […]
Hahn to lead bladder cancer program at Johns Hopkins
Noah Hahn, MD, recently concluded his tenure as chief scientific officer of the Hoosier Oncology Group and associate professor of medicine at the Indiana University Melvin and Bren Simon Cancer Center (IUSCC). Hahn joins the Johns Hopkins School of Medicine and the Sidney Kimmel Comprehensive Cancer Center as bladder cancer program director starting April 2014.
Hahn to lead bladder cancer program at Johns Hopkins
Noah Hahn, MD, recently concluded his tenure as chief scientific officer of the Hoosier Oncology Group and associate professor of medicine at the Indiana University Melvin and Bren Simon Cancer Center (IUSCC). Hahn joins the Johns Hopkins School of Medicine and the Sidney Kimmel Comprehensive Cancer Center as bladder cancer program director starting April 2014.
Hoosier Cancer Research Network Researcher to be Featured in Discovery Channel documentary
IU Simon Cancer Center breast cancer oncologist George Sledge, MD, is featured in the Discovery Channel documentary “Getting Personal: The Shifting Landscape of Cancer Care.” New research is shedding light on the genetic causes of cancer, as well as the molecular make-up of individual cancers, paving the path for more effective therapies and more personalized […]
Hoosier Cancer Research Network Researcher to be Featured in Discovery Channel documentary
IU Simon Cancer Center breast cancer oncologist George Sledge, MD, is featured in the Discovery Channel documentary “Getting Personal: The Shifting Landscape of Cancer Care.” New research is shedding light on the genetic causes of cancer, as well as the molecular make-up of individual cancers, paving the path for more effective therapies and more personalized […]
Matei receives 2011 Research Frontiers Trailblazer Award
Dr. Daniela Matei received the Research Frontiers Trailblazer Award for her discoveries related to the biology of ovarian cancer at the second annual Research Day conference. The Trailblazer award honors outstanding IUPUI researchers who are “showing great promise in becoming nationally and internationally known for their accomplishments in advancing the frontiers of knowledge.”
ASCO president and Hoosier Oncology Group investigator Dr. George Sledge testifies before president’s cancer panel

ASCO President, Dr. George W. Sledge, was among the clinician, government and industry leaders selected to testify before the President’s Cancer Panel in Boston on September 22, 2010. The meeting, titled, “The Future of Cancer Research: Accelerating Scientific Innovation,” focused on assessing the past 40 years of cancer research and exploring potential future enhancements and […]
ASCO president and Hoosier Oncology Group investigator Dr. George Sledge testifies before president’s cancer panel

ASCO President, Dr. George W. Sledge, was among the clinician, government and industry leaders selected to testify before the President’s Cancer Panel in Boston on September 22, 2010. The meeting, titled, “The Future of Cancer Research: Accelerating Scientific Innovation,” focused on assessing the past 40 years of cancer research and exploring potential future enhancements and […]
Longtime Hoosier Oncology Group investigators hold national leadership roles

George Sledge, M.D., can add yet one more title to his long list of impressive credentials and titles: President of the American Society of Clinical Oncology (ASCO). Dr. Sledge is most known for his ground-breaking work in breast cancer research, with a specific interest in the use of angiogenesis blockers, or disruption of the blood […]
Longtime Hoosier Oncology Group investigators hold national leadership roles

George Sledge, M.D., can add yet one more title to his long list of impressive credentials and titles: President of the American Society of Clinical Oncology (ASCO). Dr. Sledge is most known for his ground-breaking work in breast cancer research, with a specific interest in the use of angiogenesis blockers, or disruption of the blood […]